Picture courtesy of Bipolar androgen therapy (BAT): A patient's guide
I'm always on the lookout for new and innovative ways to treat prostate cancer. I'm especially interested in therapies that overcome the central problem of drug resistance.
As I've previously discussed, the average time for men on androgen deprivation therapy (ADT) to progress from castrate-sensitive prostate cancer (responsive to androgen deprivation therapy) to castrate-resistant prostate cancer (no longer responsive to androgen deprivation therapy) is 18-24 months. 1 2
This progression from castrate-sensitive prostate cancer (CSPC) to castrate-resistant prostate cancer (CRPC) mainly occurs due to the selective pressures of testosterone-inhibiting drugs. These pressures at the molecular level result in mutations, which make tumor cells no longer highly responsive to suppressing or blocking testosterone.
What's even more concerning is that 20-40% of men have no response to testosterone-inhibiting drugs such as enzalutamide and abiraterone.3 4 This non-response is because their tumor cells already have a built-in resistance to those drugs, known as primary resistance.
Drug resistance to prostate cancer is a significant issue of concern. That's the main reason I rail against the continued development of clinical trials that focus on the suppression and blocking of testosterone without prioritizing clinical trials that tackle drug resistance.
So when I find a therapy that not only shows effectiveness in treating CRPC but can delay and reverse drug resistance, I get excited. This therapy involves using an agent that is considered a big no-no in prostate cancer - testosterone!
Bipolar androgen therapy - a paradox
Bipolar androgen therapy (BAT) uses intermittent supraphysiologic doses of testosterone to treat castrate-resistant prostate cancer effectively. The term "bipolar androgen therapy" was coined by Samuel Denameade, M.D., who has been publishing studies on this therapy since 2014.
Dr. Denmeade is the R. Dale Hughes Professor of Oncology and Urology at the Johns Hopkins University School of Medicine and Director of the Genitourinary Oncology Division for the Johns Hopkins Kimmel Cancer Center.
He has written and published a patient's guide to BAT, which I encourage you to read, but I’ll summarize this guide in this newsletter.
What is BAT?
According to this guide, "BAT is a new treatment approach for asymptomatic men with castration‐resistant prostate cancer. BAT involves administration of sufficient amounts of testosterone to rapidly achieve a higher than normal level (i.e., supraphysiologic) of testosterone in the blood.
The normal level of testosterone in the blood of a 70‐year‐old‐man is 300–400 ng/dl. The testosterone level in a man on ADT is <50 ng/dl.
BAT is achieved by injecting a long‐lasting (depot) generic form of testosterone known as testosterone cypionate into the muscle of the buttocks every 28 days. The dose of testosterone cypionate is 400 mg. This is considered a high dose of testosterone but this dosage is within the FDA‐approved dose range of this drug.
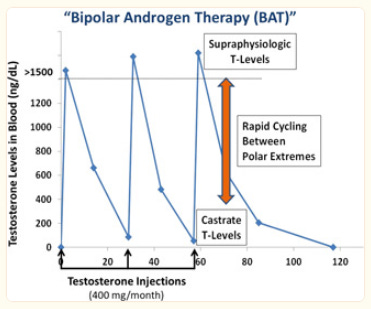
The term "bipolar androgen therapy" was coined to reflect the fact that over a 28‐day treatment cycle, the blood levels of testosterone oscillate between the polar extremes of supraphysiologic (1000–3000 ng/dl) to nearly castrate (100–200 ng/dl).
In order for this to occur, all patients should continue to receive concurrent ADT throughout treatment with BAT, and ADT should not be stopped when BAT is administered."
How does BAT work?
BAT works by:
Disrupting the normal growth cycle of the prostate cancer cell.
Producing breaks in the DNA strands of the prostate cancer cell.
Turning off essential genes that promote prostate cancer cell growth.
Activating stress pathways in the prostate cancer cell.
Stimulating the immune system in men treated.
BAT works so well in castrate-resistant prostate cancer cells because these cells have finely tuned androgen receptors (AR) as an adaptation to low testosterone levels. This fine-tuning makes these cells vulnerable to sudden exposure to super-high testosterone levels.
As Dr. Deanamide puts it, "Now it has to suddenly deal with too much androgen bound to AR. This high level “gums up the works,” so to speak. It disrupts the ability of the prostate cancer cell to divide as part of the growth cycle. In response, the prostate cancer cell either stops growing or dies."
According to Dr. Deanmide, BAT is designed to repeatedly shock the prostate cancer cells by alternating between these polar extremes of high and low testosterone levels.
“The rapid change in testosterone levels does not give prostate cancer cells sufficient time to adapt to the underlying environment because it is always changing. This method keeps the prostate cancer cells constantly guessing."
Safety of BAT
Dr. Deanamide and his team have treated approximately 350 men with BAT; most received an average of six 28-day cycles. He opines that BAT has proven safe and well‐tolerated by most men with castrate-resistant prostate cancer.
The most common side effects of BAT were mild to moderate and included:
hot flashes
breast tenderness
generalized muscle aches
swelling in the lower legs
Because testosterone can cause fluid retention, BAT should be used with caution in men with congestive heart failure. In addition, BAT can cause elevations in blood pressure.
Dr. Deanamide observed isolated incidents of heart attack and stroke in men treated with BAT, but it wasn't clear if those events were due to BAT. Regardless, BAT should be used with caution in men with cardiovascular disease.
BAT and quality of life
In studies, BAT improved the quality of life in men with castrate-resistant prostate cancer, especially regarding physical function, emotional well-being, and fatigue. Across all studies, the most significant area of improvement was with sexual function.
For many men who became impotent due to androgen deprivation therapy, BAT restored their ability to maintain an erection for suitable intercourse.
In one study, men experienced the following changes on BAT:
9% decrease in abdominal fat
12% increase in muscle mass
12% decrease in low-density lipoprotein (LDL)
27% decrease in triglycerides
9% decrease in high-density lipoprotein (HDL)
Contraindications to BAT:
Castrate-sensitive prostate cancer.
Uncontrolled bone pain due to prostate cancer.
Men who are at risk for urinary tract obstruction.
Types of responses to BAT
Dr. Deanamide sees three types of responses to BAT:
No response to BAT with PSA levels that continue to rise with signs of worsening prostate cancer on initial scans. These men come off BAT quickly, usually within three months.
PSA drops quickly with initiation of therapy - typically greater than 50%, which can last for many months. This response is frequently associated with decreasing tumor size on scans. They remain on BAT until PSA begins to rise above the starting level or until scans show the cancer growing again.
A significant increase in PSA after the first or second cycle of BAT. Then, the PSA often plateaus as a stable level or rises gradually over time. These men don't show a decrease in tumor size on scans but instead. exhibit a prolonged stabilization of disease, some lasting for several years. They are continued on BAT as long as they show a clinical benefit and their condition remains stable on imaging.
Effectiveness of BAT in metastatic castrate-resistant prostate cancer (CRPC)
Johns Hopkins has studied BAT in four clinical trials, primarily in men with CRPC who have also received treatment with enzalutamide, abiraterone, or both.
In men with CRPC progressing on either enzalutamide or abiraterone, about 33% of patients have a drop in PSA or objective response on scanning after three cycles of BAT. The duration of this response is about six months.
In one study, BAT was compared to enzalutamide in men with CRPC progressing on abiraterone. Although BAT (androgen) and enzalutamide (antiandrogen) are directly opposite therapies, the response to treatment was similar.
25% of men on either treatment had a 50% decrease in PSA lasting about six months for both treatments. 24% of men on BAT versus 4% on enzalutamide showed an objective response on scans. BAT was significantly more effective than enzalutamide in a subset of men with a short (< 6 months) response to abiraterone.
BAT reverses drug resistance
As prostate cancer cells adapt and mutate to low testosterone levels after treatment with androgen deprivation therapy, they increase the sensitivity and number of androgen receptors. When exposed to bipolar androgen therapy, the prostate cancer cells down-regulate the androgen receptors, and these men once again become sensitive to androgen-blocking drugs like enzalutamide.
In a study called RESTORE, men with CRPC whose cancers were worsening on enzalutamide. Enzalutamide was stopped, and BAT was started. Once BAT stopped working, this group received enzalutamide, and in 70%, enzalutamide started working again, lasting about six months.
Surprisingly, men who didn't respond to three cycles of BAT still responded to retreatment with enzalutamide. In other words, even though BAT didn't work in these men, they overcame their resistance to enzalutamide by giving them BAT.
For men who received abiraterone, three cycles of BAT, and then retreatment with abiraterone, the PSA response was much lower at 17% with a duration of only four months.
In one arm of the RESTORE study, a group of men with CRPC who had never received enzalutamide or abiraterone, treatment with BAT resulted in a poor PSA response of about 15%.
However, once they went on to receive enzalutamide or abiraterone, the PSA response was 95%. 85% of these men had more than a 90% decrease in PSA; in 50%, their PSA became undetectable. The duration of this PSA response lasted about 25 months.
In comparison, in the studies that led to the FDA approval of enzalutamide and abiraterone for men with CRPC, the PSA response was only 78% and 62%, respectively. The duration of the response in the studies for FDA approval was only 11 months.
In another study called TRANSFORMER, men with CRPC who received enzalutamide after abiraterone, or vice versa, the PSA response was 25%, lasting about four months. In men who received BAT first, then enzalutamide, the PSA response was about 80%, lasting about 11 months. BAT improved overall survival by eight months.
These findings point to the potential for BAT to improve the response to subsequent androgen-blocking therapies once men become castrate-resistant. The combined results from the RESTORE and TRANSFORMER studies also show that BAT is highly effective at restoring sensitivity and disrupting resistance to antiandrogens like enzalutamide.
BAT clinical pearls
BAT is given as an intramuscular injection of testosterone cypionate every 28 days.
BAT should only be given to patients with castrate‐resistant (NOT hormone‐sensitive) prostate cancer.
BAT should NOT be given to prostate cancer patients with cancer‐related bone pain.
BAT should not be given to patients with urinary obstruction due to enlarged prostate or prostate cancer.
BAT should be combined with ongoing ADT or surgical castration.
BAT may be continued despite PSA a rising PSA if clinical benefit and stable scans show no progression.
BAT may render CRPC patients sensitive to enzalutamide or abiraterone after ADT or after prior progression on these drugs
BAT should preferentially be administered in a clinical trial, especially when used with other experimental therapies.
The current state of BAT
I came across an excellent video discussing BAT titled "The State of Bipolar Androgen Therapy in Prostate Cancer - Emmanuel Antonarakis." In it, the President and Chief Executive Officer of The Prostate Cancer Foundation (PCF), Charles J. Ryan, M.D., discusses BAT with Emmanuel Antonarakis, M.D., Clark Endowed Professor of Medicine, Division of Hematology, Oncology and Transplantation, University of Minnesota.
Dr. Antonarakis published several clinical studies with Dr. Deanmeade, and in this video, he discusses that men with prostate cancer who harbor germline BRCA2 and ATM mutations and men with somatic P53 mutations appear to be preferentially sensitive to BAT.
These are significant findings, as men with these types of mutations have a worse prognosis.
We need more physicians and researchers like Dr. Deanmeade and Dr. Antonarakis who think "out of the box" and research therapies that not only improve overall survival in men with prostate cancer but also tackle drug resistance.
Until next time, stay well.
Much love,
Keith
Hamberg P, Verhagen PC, de Wit R. When to start cytotoxic therapy in asymptomatic patients with hormone refractory prostate cancer? Eur J Cancer. 2008;44(9):1193–1197.
Sternberg CN. Systemic chemotherapy and new experimental approaches in the treatment of metastatic prostate cancer. Ann Oncol. 2008;19(Suppl 7):vii91–vii95.
Scher HI, Fizazi K, Saad F, et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N Engl J Med 2012;367:1187-1197
Ryan CJ, Smith MR, de Bono JS, et al. Abiraterone in metastatic prostate cancer without previous chemotherapy. N Engl J Med 2013;368:138-148



That was the most comprehensive and best explanation I have read to date about BAT. Consistent with your post, rather than characterize BAT as “paradoxical “, I would describe it as further evidence that continuing to “feed” the cancer cells with testosterone during the entire treatment protocol will lessen the risk, or extend the time, that the cells become castration resistant. I have used intermittent anti androgen therapy, plus metastasis directed therapy, to remain metastatic hormone sensitive for more than 15 years. By skipping the LHRH agonist or antagonist and using only ant- androgen mono therapy, testosterone rises during the on period, thereby feeding the cells with testosterone during the off period(“BAT Lite”). This lessens the need for the cells to mutate to find lower and lower levels of testosterone. The potential benefits of this protocol is described well by Dr. Gatenby at Moffitt Cancer Center in Tampa (the Darwinian or evolutionary approach to cancer treatment).