Positron emission tomography (PET) scans combine radioactive tracers with computed tomography (CT) or magnetic resonance imaging (MRI) scans to assess for metabolically active cancer in the body. CT and MRI scans show the detailed anatomy of the body, while PET tracers show the molecular changes of cancer occurring in the tissues.
To determine which PET scan works best in what instance, you have to consider its sensitivity and specificity. Sensitivity represents the ability of the scan to identify true positive cases correctly.
Specificity measures how well a test can correctly identify people who do not have a specific condition or disease. If a test has high specificity, it can exclude most people who don't have the condition.
National Comprehensive Cancer Network (NCCN)
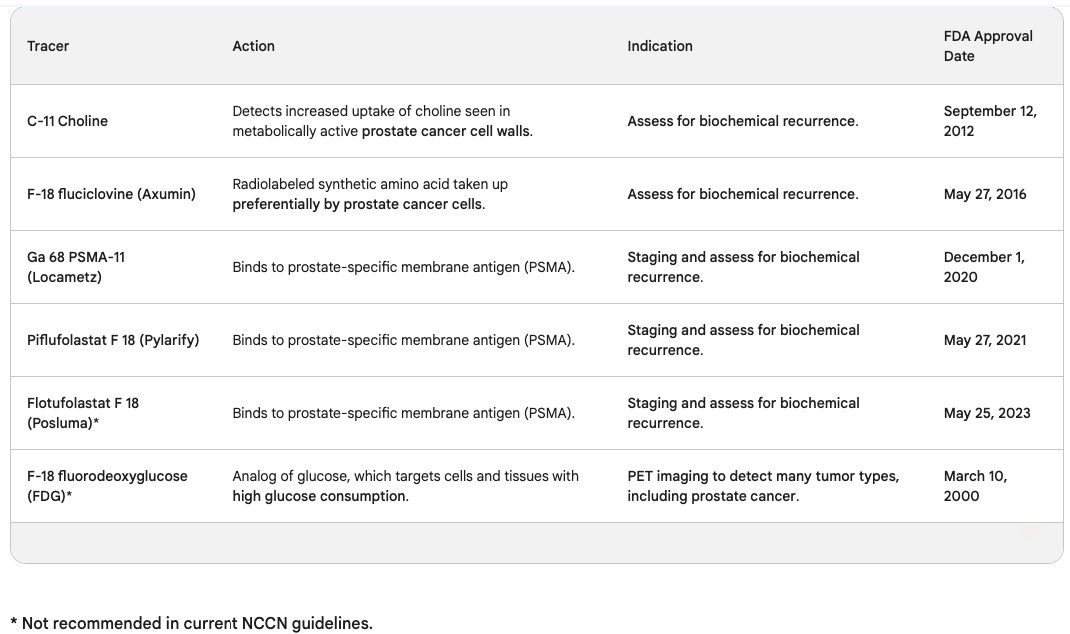
The United States Food and Drug Administration (FDA) approves PET tracers for certain cancers and circumstances. This newsletter discusses six PET tracers for prostate cancer mentioned in the National Comprehensive Cancer Network (NCCN) guidelines for prostate cancer.
The NCCN is an alliance of thirty-three leading cancer centers in the United States that create oncology clinical practice guidelines.
These guidelines are "the recognized standard or clinical direction and policy in cancer care and are the most thorough and frequently updated clinical practice guidelines available in any area of medicine."
Positron emission tomography (PET) scans for prostate cancer
The FDA approved the C-11 Choline PET tracer on September 12, 2012. Choline is an essential element of cell walls, and this scan detects the increased uptake of choline seen in metabolically active prostate cancer cell walls. A C-11 Choline PET scan is for imaging men with biochemically recurrent prostate cancer.
Biochemical recurrent prostate cancer means the prostate-specific antigen (PSA) has risen to a certain level after prostate cancer treatment.
The FDA approved F-18 fluciclovine, brand name Axumin, on May 27, 2016. It is a radiolabeled synthetic amino acid taken up preferentially by prostate cancer cells. Its main indication is also for biochemically recurrent prostate cancer. This PET scan produced a false-positive result for me, which I discussed in newsletter 021.
The four prostate-specific membrane antigen (PSMA)-based PET tracers are:
Ga 68 PSMA-11, brand name Locametz, FDA approved December 1, 2020
Piflufolastat F 18, brand name Pylarify, FDA approved May 27, 2021
Ga 68 gozetotide, brand name Illuccix, FDA approved December 17, 2021.
Flotufolastat F 18, brand name Posluma, FDA approved May 25, 2023
PSMA-based PET tracers are for the initial diagnosis and staging of prostate cancer patients with suspected metastases and imaging patients with suspected biochemical recurrence.
Per Prostate Cancer-PET Imaging Update, Ga-68 PSMA-11 was found to be significantly better at detecting prostate cancer recurrence than Axumin in patients with early biochemical recurrence after prostatectomy, with the exception being locoregional recurrence, where Axumin was more sensitive due to its lack of radiotracer in the bladder.
For more information about the mechanism of action of PSMA-based PET scans and how this technology allows for diagnosing and treating advanced prostate cancer, please go to newsletter 012.
In addition, The NCCN guidelines indicate that both Ga-68 PSMA-11 and F-18 piflufolastat PSMA imaging are eligible to determine whether men are candidates for treatment with Lutetium-177-PSMA-617 radioligand therapy.
Flotufolastat F 18, brand name Posluma, is not mentioned in the current NCCN guidelines, mainly because its approval by the FDA was so recent.
The PET tracer F-18 fluorodeoxyglucose (FDG) tracer is an analog of glucose, which targets cells and tissues with high glucose consumption. The FDA approved it to detect many tumor types, but the NCCN guidelines recommend not using it to stage prostate cancer.
This recommendation is because prostate cancer is usually slow-growing and typically doesn't uptake glucose (sugar) to the degree other types of malignant cells do.
In addition, an FDG PET scan can't tell the difference between prostate cancer and inflammation. So if someone has an elevated prostate-specific antigen (PSA) due to prostatitis, the FDG PET scan may result in false-positive imaging. In this case, false-positive imaging means the radiologist might interpret the scan as cancer, but it is abnormal due to prostatitis.
Current NCCN guidelines
The current guidelines say, "C-11 choline or F-18 fluciclovine PET/CT or PET/MRI are good at detecting small-volume recurrent disease in soft tissues and bone. But studies suggest that PSMA PET imaging has a higher sensitivity than C-11 choline and F-18 fluciclovine PET imaging, especially at very low PSA levels."
The NCCN guidelines go on to say, "Because of the increased sensitivity and specificity of PSMA PET tracers for detecting micrometastatic disease compared to conventional imaging (CT/MRI/bone scan), at both initial staging and biochemical recurrence, PSMA PET/CT and PSMA PET/MRI can serve as equally effective, if not more effective front-line imaging tools in prostate cancer."
PSMA PET scan controversy
PSMA PET/CT and PSMA PET/MRI scans are now the preferred scans for staging and detecting biochemically recurrent prostate cancer by most practitioners with access to these scans. Just like with proton beam therapy centers, only some men in the United States have easy geographical access to this type of technology.
As with any new technology, there is controversy. Some practitioners, especially those associated with academic teaching centers, ironically think PSMA PET scans are too accurate for staging prostate cancer. In other words, PSMA PET scans will detect small areas of metastatic disease that conventional scanning - plain CTs and bone scans - would not.
They base this thought on the fact that most clinical trials have used conventional imaging to determine treatment for prostate cancer at various stages. And because of that, these practitioners think that prostate cancer will now be "over-staged," leading to earlier treatments. They are concerned that earlier treatment for many of these men won't lead to increased overall survival but will result in a poorer quality of life.
Does this argument sound familiar? It's because it's similar to the concerns about the overuse of PSA screening leading to unnecessary treatment of prostate cancer, resulting in a poorer quality of life for some men.
The practitioners voicing these concerns say we should wait for more high-quality clinical trials with PSMA PET scanning to determine when to best use these more accurate scans. Other practitioners note that earlier detection of metastatic disease will prevent unnecessary prostatectomies and their associated morbidities.
The advent of PSMA PET scanning is another reason why seeing a specialist at a prostate cancer center of excellence is so important.
The usefulness of F-18 fluorodeoxyglucose (FDG) PET scans
The FDG PET scan for prostate cancer is frowned upon by many practitioners who treat prostate cancer. The NCCN guidelines specifically say not to use it, but the FDG PET scan does have a place in evaluating and managing advanced prostate cancer.
The Evolving Role of 18F-FDG PET/CT in Diagnosis and Prognosis Prediction in Progressive Prostate Cancer says, "18F-FDG PET/CT is shown to be a useful prognostic tool in selected patients with advanced prostate cancer."
It also says, "Intraprostatic uptake of 18F-FDG imaged by PET/CT suggests (that) aggressive behavior and castration resistance with increased glucose uptake." And "clinical data have shown that FDG uptake tends to increase in more aggressive prostate cancer, either recurrent or metastatic."
This article indicates that in advanced prostate cancer, uptake of FDG indicates a more aggressive cancer. Some castrate-resistant prostate tumors that didn't express PSMA and had a high uptake of FDG turned out to be neuroendocrine-type prostate cancers. As discussed in a prior newsletter, neuroendocrine prostate cancers are extremely aggressive.
Another way researchers are using FDG PET scans is to determine who might benefit more from PSMA radioligand therapy and chemotherapy. In the TheraP trial, men with a high metabolic tumor volume on FDG scanning had lower responses to both types of therapy.
The answer
If most prostate cancer specialists were asked, "What is the best PET scan for CT or MRI scanning in prostate cancer?" They'd probably answer PSMA PET. But in reality, the true answer is, "It depends."
If you have any topics in prostate cancer you'd like me to cover, please place those topics in the comments section. Until next time, stay cool, and lots of love!
Keith



Is radiation the answer for a 70 year old whose prostate cancer barely made it to the intermediate stage?