Beware of Certain Clinical Trials - 039
Are pharmaceutical companies designing unethical clinical trials?
Before I discuss the topic of this newsletter, I want to follow up on newsletter 036, in which I discussed the health insurance issues related to choosing the site where I underwent my last positron emission tomography (PET) contrasted tomography (CT) scan.
To good to be true
I wrote that I chose Precision Imaging in Jacksonville for the scan due to the low out-of-pocket price they quoted me based on my health insurance. Do you know that old saying about something is too good to be true? Well, that was the case here.
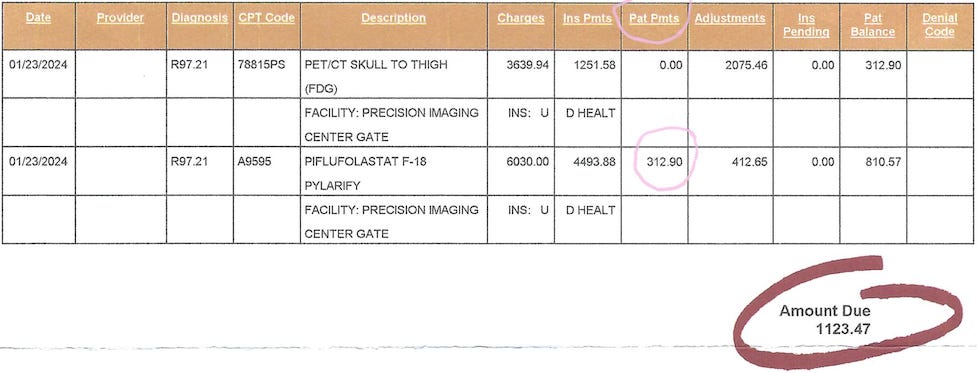
The billing department quoted me $312.90 for the out-of-pocket cost of the PET CT scan portion, not the radioactive tracer. After receiving a bill from Precision Imaging one week later, it showed I owed them an additional $1123.47.
What's strange is that they had a chance to catch this error on the day of my scan when I asked to pay them my total out-of-pocket amount due with a credit card. They charged my card for $312.90.
Phone tag
I called the billing department and spoke with a representative who informed me that my quote was only for the PET CT portion. She couldn't explain why this error occurred and recommended I speak with the billing manager. She transferred me, and the phone call went directly to her voicemail, so I left a message.
A few hours later, the billing manager called me back, but I couldn't take the call because I was at work. She left a voice message saying she had gotten a message that I needed to speak with her. I called back and again left a message on her voicemail.
The power of a Google review
After giving her five days to return my call, I called her again and left another voicemail. After two more days without a callback, I left one-star reviews on Google Reviews and Yelp with a short discussion of what happened.
About an hour later, I received an email that someone had responded to my Google review. I checked, and the Chief Executive Officer (CEO) of Precision Imaging responded by leaving his email address and asking me to please email him about this.
I emailed him and included my phone number. The following day, the billing manager called and politely apologized for what had happened. She said it was a mistake on their part and that she had taken corrective action so that it wouldn't happen to another patient.
She also offered to let me pay half of the $1123.47 as recompense. I thanked her and told her I had already deleted the reviews because her CEO had contacted me to help resolve this issue. So, I paid $873.90 instead of $312.90 for the Pylarify scan.
Surprisingly, the CEO called me the next day to ensure everything had worked out okay. He was very kind and apologetic. I was impressed and will continue using and recommending Precision Imaging for radiologic testing.
Disturbing pattern in clinical trial design for prostate cancer
Now, on to today's topic, which concerns a pattern developing in clinical trial designs for prostate cancer that needs to stop. This pattern involves designing clinical trials with suboptimal control arms.
A typical randomized clinical trial is a prospective study that measures the effectiveness of a new intervention or treatment compared to a control. A computer randomly assigns patients to the treatment or control arms. Randomization balances the two arms to provide an equal number of comparable patients in each arm.
Patients in the treatment arm receive the new intervention or treatment. Patients in the control arm receive either the standard of care treatment, no treatment, or a placebo. However, according to Vinay Prasad, M.D., M.P.H., a hematologist oncologist who studies clinical trial designs in oncology,
"The control arm should receive the best available drugs."
Giving no treatment or a placebo is not an acceptable option in cancer trials where the patient has a life-threatening disease. In addition, he emphasizes that it is unethical if the patients in the control arm don't get the best standard of care therapies known at that time.
Substandard control arms
In recent clinical trials of men with metastatic castrate-resistant prostate cancer (mCRPC) it appears that designers are creating substandard control arms. One would hope this design quirk is pure ignorance, but creating substandard control arms can make the treatment arm outcomes falsely favorable.
Pharmaceutical companies fund and help design the majority of clinical trials for mCRPC. Surely, “big pharma” wouldn't risk men's lives in the control arm to help make their drug or other treatment look better than it is.
After all, making their treatment look better has the potential to get it approved by the United States Food and Drug Administration (FDA), which results in mind-boggling revenue for pharmaceutical companies. Verified Market Research reports that the metastatic castrate-resistant prostate cancer treatment market was 13.4 billion dollars in 2023.
Uncovering problematic clinical trials
A Journal of Clinical Oncology article and Dr. Prasad discuss a recently published clinical trial in mCRPC with a substandard control arm.
The PROfound trial was a phase 3 randomized clinical trial that evaluated the effectiveness of olaparib, a poly (ADP-ribose) polymerase (PARP) inhibitor, versus physician's choice of enzalutamide or abiraterone in patients with mCRPC and mutations in specific DNA repair genes.
Medical writers funded by the pharmaceutical company wrote the trial manuscript.
Patients were randomized 2:1 to receive either olaparib (treatment arm) or the physician's choice of enzalutamide or abiraterone (control arm).
The control arm was labeled "physician's choice" but was limited to only enzalutamide or abiraterone, which were treatments most patients had already progressed on before entering the clinical trial.
45% of patients had already gotten enzalutamide.
35-40% of patients had already gotten abiraterone.
Phase 2 data show that the response rate for sequencing abiraterone followed by enzalutamide is only 30%, and for enzalutamide followed by abiraterone, a pitiful 5-7%.
Clinical trials show that sequencing abiraterone and enzalutamide does not improve overall survival.
20% of patients in the control arm had already failed both drugs making the drugs for them a placebo with adverse side effects.
80% of patients in the control arm were denied access to effective chemotherapies like docetaxel or cabazitaxel, which have proven survival benefits in mCRPC.
The trial reported an overall survival benefit for olaparib in the cohort with BRCA1/2 or ATM gene mutations (Cohort A).
However, by limiting control arm options, the trial may have overestimated the benefit of olaparib.
The journal article revealed another mCRPC clinical trial with a suboptimal control arm. The VISION trial evaluated the efficacy of 177Lu-PSMA-617 radioligand therapy plus standard of care versus standard of care alone in patients with advanced prostate-specific membrane antigen (PSMA) positive mCRPC who had previously received one or more androgen receptor targeting agents (ARTA), including abiraterone and enzalutamide, and one to two taxane chemotherapy regimens.
The trial reported prolonged imaging-based progression-free survival (PFS) and overall survival (OS) with 177Lu-PSMA-617. However, the trial design had significant limitations:
Almost 60% of enrolled patients had received prior docetaxel chemotherapy but not cabazitaxel, a second-line taxane chemotherapy with proven OS benefit.
The protocol prohibited chemotherapy in the standard of care control arm. As a result, the control arm mainly consisted of steroids (60%) and ARTA after ARTA (60%), even though 40% of patients had already received 2 ARTAs and 8% had received >2 ARTAs before enrollment.
This control arm was substandard because a substantial proportion of patients, especially the 60% who had not received cabazitaxel, would have been eligible for and benefited from this life-prolonging therapy.
28% of patients received subsequent therapy after progression, including 19% who received a taxane, indicating more patients could have received cabazitaxel at enrollment.
Only 17% of patients had received Radium-223 before enrollment, another life-prolonging treatment that could have served as a more adequate control in patients with bone-predominant disease.
While the VISION trial demonstrated an overall survival benefit of four months for 177Lu-PSMA-617, this survival benefit may be overestimated because the trial design limited the control arm options. A more appropriate control arm would have allowed therapies like cabazitaxel and Radium-223 that have proven overall survival benefits.
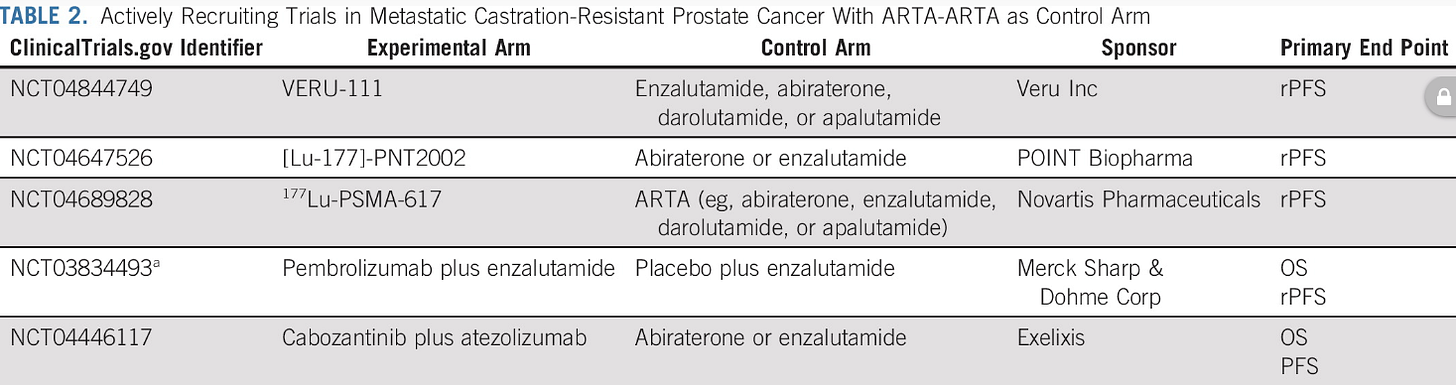
In evaluating the prevalence of substandard control arms in randomized phase 3 clinical trials for mCRPC, the researchers found five actively recruiting trials using ARTA drugs in sequence as their control arm. And guess what? Only pharmaceutical company-sponsored trials use this type of suboptimal control arm.
If you are considering participating in a clinical trial for mCRPC, ask if the control arm uses actual standard of care therapies.
Beware of clinical trials that allow the sequencing of androgen targeting (ARTA) drugs, including the following:
abiraterone acetate (Zytiga)
enzalutamide (Xtandi)
darolutamide (Nubeqa)
aspalutamide (Erleada)
Using suboptimal control arms in clinical trials raises serious ethical and clinical concerns. It undermines the trust and safety of trial participants and can lead to the approval of treatments without proven superiority over the established standard of care.
Ensuring that control arms consistently represent the best current standard of care is essential to protecting patients and maintaining the integrity of the clinical trial process.
Until next time, stay well.
Much love,
Keith